By Philippa Johnson
Review of: Daniel Hulsey, Kevin Zumwalt, Luca Mazzucato, David A. McCormick, Santiago Jaramillo. Decision-making dynamics are predicted by arousal and uninstructed movements. bioRxiv, doi: https://doi.org/10.1101/2023.03.02.530651
In a recent lab meeting, we discussed a preprint by Hulsey et al. (2023), which investigates whether fluctuations in arousal can account for some of the variability found in decision-making behaviour. In psychology experiments, we usually implicitly assume that people/animals perform the same cognitive task with the same strategy during an experimental ‘session’ – but more and more research shows that this is a bit naïve, and that we can draw false conclusions if we don’t take these nonstationarities into account. After all, the way we process information at the beginning of a boring lecture is surely very different to how we process information towards the end!
A previous paper by Ashwood et al. (2022) found that mice performing a perceptual decision making task employ a variety of different strategies over the course of testing sessions. These strategies were described by a Hidden Markov Model (HMM), in which a set of unobservable, discrete states with Markov property are inferred based on observable behaviour. Each state, or behavioural strategy, is characterised by a sigmoidal mapping between stimulus features (current stimulus, previous error/correct, previous choice and a bias term) and response (left or right; i.e., a state-specific generalised linear model). In the present paper, Hulsey and co-authors investigate whether these state switches are related to fluctuations in arousal, measured by pupil dilation, face movement and locomotion in an auditory and a visual stimulus discrimination task. Mice reported whether a Gabor was closer to a vertical or horizontal orientation, or whether an auditory tone cloud was in a higher or lower octave. They completed this task in conditions which facilitated large fluctuations in arousal, as they could run on a wheel as desired, but there were long (3-7s) inter-trial intervals.
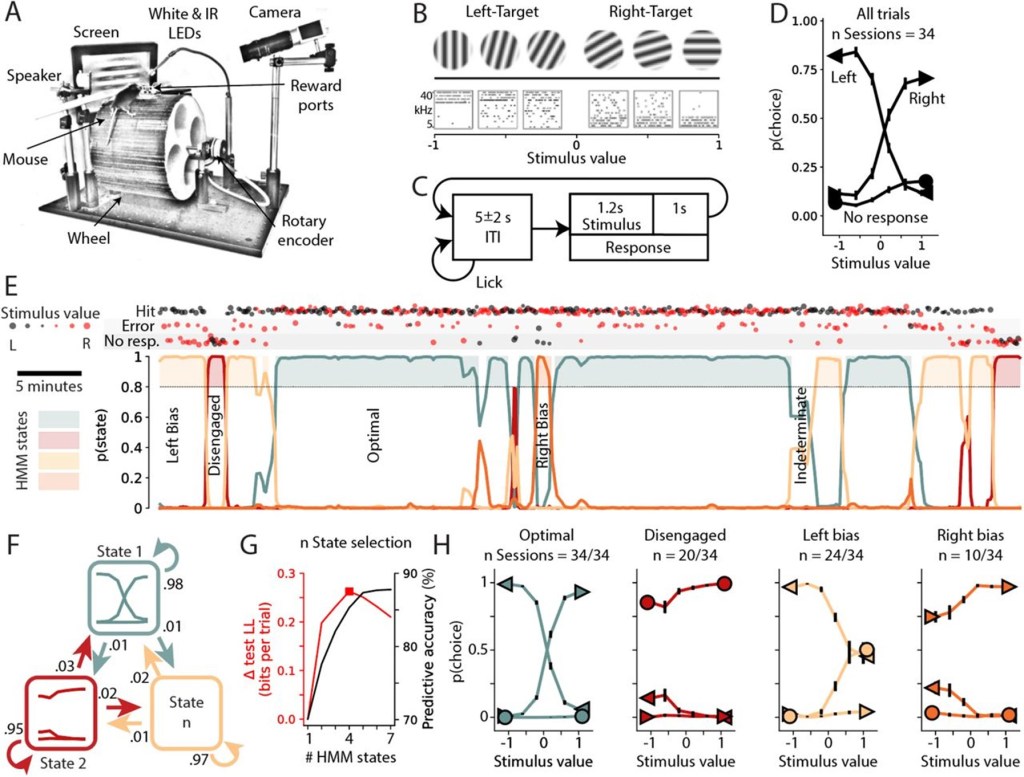
Reprinted from Figure 1 of Hulsey et al. (2023) under a CC-BY-NC-ND 4.0 licence. Here, you can see the experimental setup and design of the auditory and visual tasks, as well as GLM-HMM posterior probability time courses and psychometric curves for each of the identified states in an example animal.
The authors estimated a GLM-HMM, but with some novel variations. Firstly, the model fits were performed on data from individual mice (with concatenated sessions), without an initial global fit as is used in Ashwood et al.. We imagine this is because Hulsey et al. had more trials per animal, but we would have appreciated more information about how many sessions/trials are necessary for reliable model fits, as well as some discussion of the impact of concatenating sessions on state switches (do session start/end get picked up by the model as state switches? Does it make sense to say that a mouse is in the same state across sessions?). Secondly, no-response trials were included in the GLM by using a multinomial as opposed to Bernoulli emission. This allowed the identification of more states, namely avoid left and avoid right, and means that no-response trials (which are often found in significant numbers) are not excluded when establishing the temporal structure of task engagement. Figure 1E shows a nice visualisation of this temporal structure within one session.
This paper, combined with the results reported by Ashwood et al., really drives home the importance of considering fluctuations in engagement when analysing behavioural (and neural) data. Psychometric curves and GLM weights show that animals are using very different strategies to respond to the same stimulus and producing very different behaviour across states. In this paper, only 50% occupancy of the optimal state was found (Figure 2B). This potentially cracks the foundation of other behavioural experiments, in which it is typically assumed that animals are engaged in the task. Selectively analysing trials in which an animal/person is in an engaged state could increase power to discover relevant neural computations, by decreasing noise associated with task-irrelevant processing in disengaged states.
Additionally, this temporal segmentation of trials allows investigation of the drivers and characteristics of task (dis)engagement: the paper further investigated the relationship between the identified engagement states and arousal, measured by deviation from maximum pupil size or a movement index which is produced by combining face motion energy and locomotion speed. Hulsey et al. found that the engaged state was characterised by a median level of arousal and decreased variability in arousal indices, while disengaged states were characterised by a higher probability of high or low arousal and increased variability in arousal indices (Figure 3E & F). We noticed in Figure S2 that the U-shaped relationship between pupil diameter and probability of being in the disengaged state (red line) seems to only be present when the results from the auditory and visual tasks are combined; the disengaged states seem to be associated with a large pupil size in the auditory task and a small pupil size in the visual task. In general, we would have appreciated more discussion of the differences between the two tasks. For example, in the visual task, the pupil also responds to changes in the visual stimulation.
Most interestingly to us, Hulsey et al. found that a shift into the optimal ‘engaged’ state was preceded by a decrease in variability of arousal indices, while a shift out of this state was associated with an increase (Figure 3G). This variability was established using an average of standard deviations over the preceding 10 trials. We were curious whether one could establish how far in advance a state switch can be predicted with a more temporally precise measure, for example moment-to-moment fluctuations in pupil size. Regardless, this preceding shift in arousal has some interesting implications: perhaps a shift in arousal has a causal role in triggering shifts in engagement state. This could be achieved through noradrenaline acting as a global signal which resets the network dynamics underlying behaviour (Bouret & Sara, 2005; Karlsson et al., 2012). Alternatively, these findings could signal that behavioural states are in fact not discrete but could better be described by a model that drifts continuously from state to state.
Overall, this paper provides an interesting insight into the relationship between arousal and task engagement. The results suggest that arousal fluctuates with engagement, with median arousal levels being associated yet again with optimal performance (Yerkes & Dodson, 1908). This complements other recent investigations of fluctuations in arousal during behavioural tasks, for example, de Gee et al. (2022), who found that mice regulate their arousal to take advantage of high task utility.
References
- Ashwood, Z. C., Roy, N. A., Stone, I. R., Urai, A. E., Churchland, A. K., Pouget, A., & Pillow, J. W. (2022). Mice alternate between discrete strategies during perceptual decision-making. Nature Neuroscience, 25(2), Article 2. https://doi.org/10.1038/s41593-021-01007-z
- Bouret, S., & Sara, S. J. (2005). Network reset: A simplified overarching theory of locus coeruleus noradrenaline function. Trends in Neurosciences, 28(11), 574–582. https://doi.org/10.1016/j.tins.2005.09.002
- de Gee, J. W., Mridha, Z., Hudson, M., Shi, Y., Ramsaywak, H., Smith, S., Karediya, N., Thompson, M., Jaspe, K., Jiang, H., Zhang, W., & McGinley, M. J. (2022). Strategic self-control of arousal boosts sustained attention (p. 2022.03.04.482962). bioRxiv. https://doi.org/10.1101/2022.03.04.482962
- Hulsey, D., Zumwalt, K., Mazzucato, L., McCormick, D. A., & Jaramillo, S. (2023). Transitions between discrete performance states in auditory and visual tasks are predicted by arousal and uninstructed movements (p. 2023.03.02.530651). bioRxiv. https://doi.org/10.1101/2023.03.02.530651
- Karlsson, M. P., Tervo, D. G. R., & Karpova, A. Y. (2012). Network Resets in Medial Prefrontal Cortex Mark the Onset of Behavioral Uncertainty. Science, 338(6103), 135–139. https://doi.org/10.1126/science.1226518
- Yerkes, R. M., & Dodson, J. D. (1908). The relation of strength of stimulus to rapidity of habit-formation. Journal of Comparative Neurology and Psychology, 18(5), 459–482. https://doi.org/10.1002/cne.920180503